A UC Santa Cruz special report: Revealing the Ribosome

From antibiotics to the origins of life, Harry Noller's findings on the structure of the ribosome and how it carries out protein synthesis have broad implications
After devoting 50 years of his life to one fundamental problem at the heart of molecular biology, Harry Noller is now basking in the recognition that comes with the 2017 Breakthrough Prize in Life Sciences. The $3 million prize honors Noller, the Robert L. Sinsheimer Professor of Molecular Biology at UC Santa Cruz, for revealing how the complex molecular machines called ribosomes build the proteins in all living cells.
“It’s been a long haul, but it’s been exciting all the way,” said Noller, who directs the Center for Molecular Biology of RNA at UC Santa Cruz.
Proteins of all sizes and shapes do most of the work in cells, and the recipes for making those proteins are written in the DNA sequences of genes. Ribosomes perform the crucial task of reading the genetic instructions and synthesizing the specified proteins. Inside every living cell, tens of thousands of them churn out proteins with mind-boggling speed and precision.
“The ribosome is responsible for translation between the language of DNA and RNA and the language of proteins,” Noller said. “It’s one of the most fundamental processes of life.”
Noller’s research has revealed the structure of the ribosome and how it works, pointing unexpectedly to the functional role of RNA. These results have important practical applications. Many of the most effective antibiotics work by targeting bacterial ribosomes, and findings by Noller and others have led to the development of novel antibiotics that hold promise for fighting drug-resistant infections. An estimated 2 million people in the United States are infected with antibiotic-resistant bacteria every year, and at least 23,000 people die each year as a direct result of these infections. As new strains of antibiotic-resistant bacteria continue to emerge, the need for new antibiotics is becoming increasingly desperate.
The caveman and the Ferrari
Noller began studying the ribosome in 1966 as a postdoctoral researcher in Geneva, Switzerland. He has compared himself to a caveman who finds a Ferrari parked outside his cave and starts trying to figure out how it works. Once he and his buddies discover the ignition, the steering, the accelerator, and the brakes, they think they’ve got it all figured out. Then someone pops the hood, and they look at the engine and realize they haven’t even begun to understand the Ferrari.
“That’s where we were with the ribosome,” Noller said. “We knew what it did, but we didn’t know how it worked.”

As a biomolecular structure, the ribosome is enormously complex, comprised of proteins and RNA molecules intricately folded and intertwined. The bacterial ribosomes studied in Noller’s lab are made up of three large RNA molecules and more than 50 different proteins.
When Noller came to UC Santa Cruz in 1968, he started trying to figure out which of the proteins performed the key functions of the ribosome. Like other molecular biologists, he assumed the proteins were the active components, while the RNA molecules provided a structural framework to hold the proteins in place. But as he systematically modified the proteins to block their activity, he found that the ribosome continued to function without them.
Finally, Noller assigned Brad Chaires, an undergraduate working in his lab, to treat ribosomes with a chemical that would modify the RNA. The results were a complete surprise. The treatment had knocked out the activity of the ribosome for the first time, not by inactivating a protein but by inactivating the RNA component.
“That was the beginning of showing that the function of the ribosome is not based on protein, it’s based on RNA. The only problem was, it was so unexpected that nobody believed it,” Noller said.
When he published those results in 1972, Noller was still an untenured assistant professor. His proposal that RNA was the active component of the ribosome was dismissed by most researchers in the field. “It was a completely heterodox view when we first proposed it, but it is now the accepted paradigm,” he said.
The discovery in the 1980s of RNA molecules that catalyze biochemical reactions (called ribozymes) helped Noller’s idea gain acceptance. In order to truly understand how the ribosome worked, however, it was essential to get a detailed picture of its molecular structure. The standard procedure for this is x-ray crystallography, which requires pure crystals of the molecular structure to be analyzed. Crystallizing something as large as a ribosome is extremely difficult, and many research groups spent decades working to determine its structure.
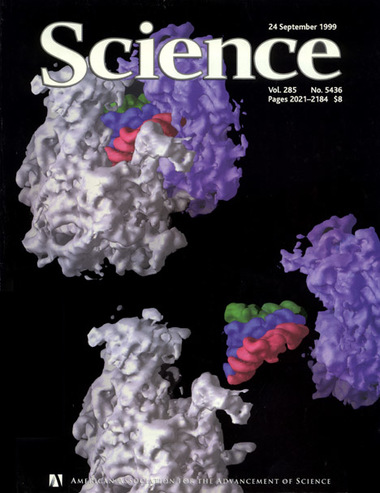
Noller’s laboratory achieved a breakthrough in 1999, producing the first high-resolution image of the molecular structure of a complete ribosome. At the time, it was the largest structure ever solved by x-ray crystallography. Noller’s team later achieved even higher-resolution images, showing details never seen before and suggesting how certain parts of the ribosomal RNA move during protein synthesis.
Translating the code
To make a new protein, the genetic instructions are first copied from the DNA sequence of a gene to a messenger RNA molecule. The ribosome then translates the sequence of the messenger RNA by matching each three-letter “codon” of genetic code with a specific protein building block, one of 20 amino acids. In this way, the ribosome builds a protein molecule with the exact sequence of amino acids specified by the gene.
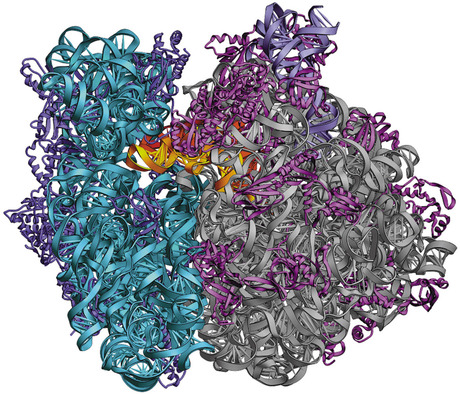
The matching of codons to amino acids is done via transfer RNA molecules, each of which carries a specific amino acid to the ribosome and lines it up with the matching codon on the messenger RNA. Images obtained by Noller’s group show the complete ribosome with a messenger RNA and two full-length transfer RNAs bound to it. Furthermore, his team has managed to freeze the ribosome in action at different stages in the process of translation, yielding a series of snapshots of the molecular machine at work.
These snapshots are like frames in a movie showing the movements of the ribosome and the central role of ribosomal RNA. They also reveal the mechanism that ensures the accuracy of the translation from genetic code to protein.
“The big question has been to understand how messenger RNA and transfer RNA are moved synchronously through the ribosome as the messenger RNA is translated into protein,” Noller said. “The transfer RNAs are large macromolecules, and the ribosome has moving parts that enable it to move them through quickly and accurately at a rate of 20 per second.”
Origins of life
The centrality of RNA to the functioning of the ribosome has helped solve a conundrum in the molecular evolution of life. If proteins are the active molecules, and proteins are needed to make new proteins, how did the first ones come to be? One solution would be if the first ribosomes were made entirely of RNA. “When we saw the structure for the first time, we saw that this was possible,” Noller said.
The realization that RNA molecules can encode genetic information, make copies, and catalyze biochemical reactions led to the idea that life began in an RNA world, with self-replicating RNA molecules preceding the evolution of DNA and proteins. Researchers are now busy working out how RNA could have arisen from nonliving chemicals in the early Earth.
“Perhaps the ultimate question of science is, Where do we come from?”Noller said. “What seemed like a miracle to everyone, including all but the most daring scientists 50 years ago, looks quite plausible now as a scenario for how you start with inorganic chemicals and arrive at living systems.”
When the 2009 Nobel Prize in Chemistry was given to three scientists for their work on the structure of the ribosome, many in the field thought Noller should have shared in that honor. But the Nobel Committee’s rules limit each prize to no more than three recipients. UCSC Chancellor George Blumenthal is among those who now feel Noller is finally getting due recognition for his extraordinary achievements.
“Harry Noller has performed a tour de force in molecular biology for the past 50 years,” Blumenthal said. “This recognition is well-deserved.”
